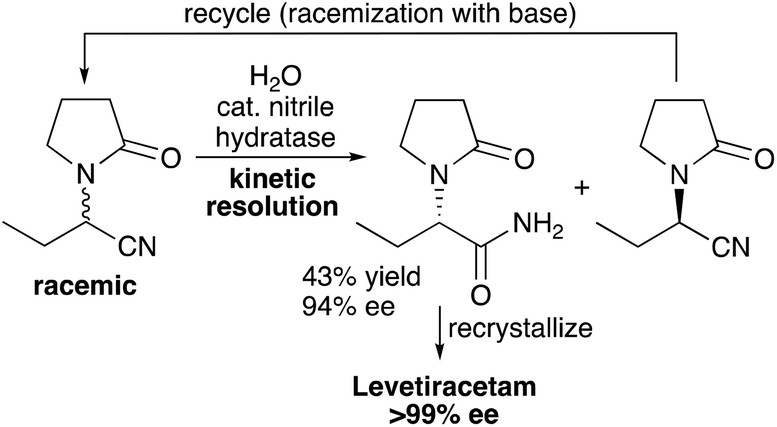
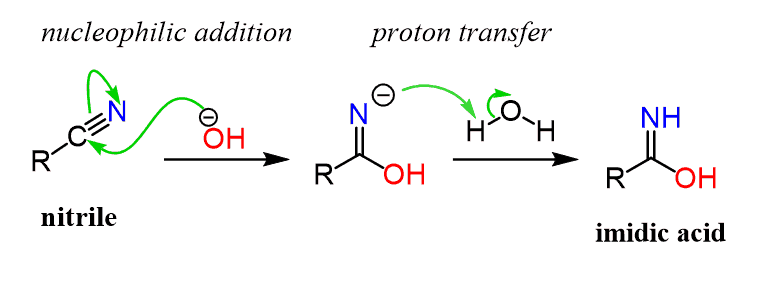
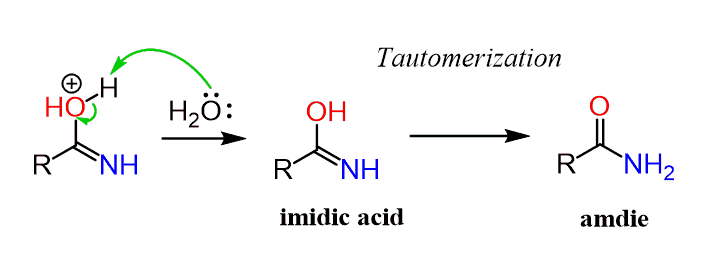
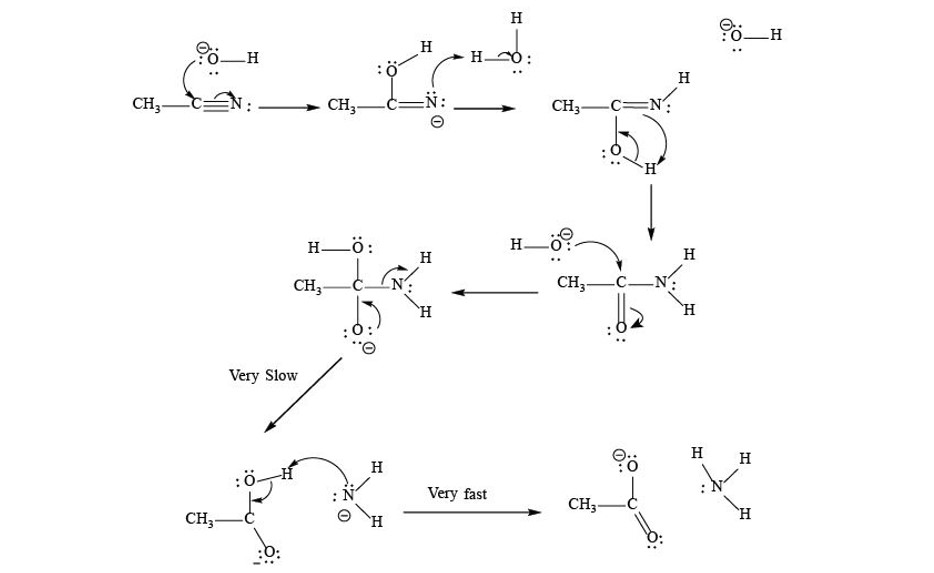
Hydrogen Acceptor‐ and Base‐Free N‐Formylation of Nitriles and Amines using Methanol as C1 Source - Kang - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library
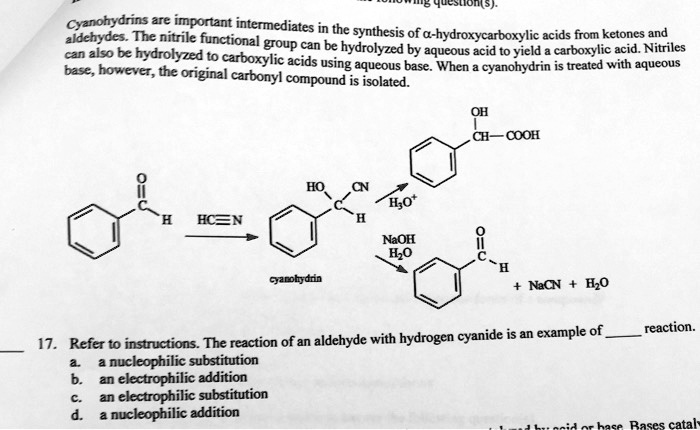
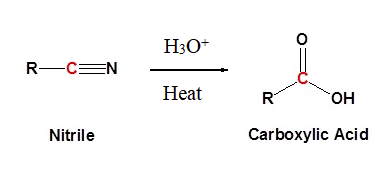
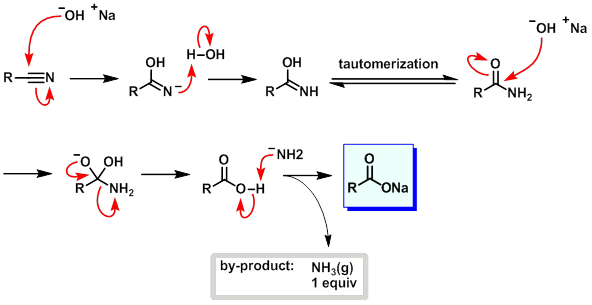
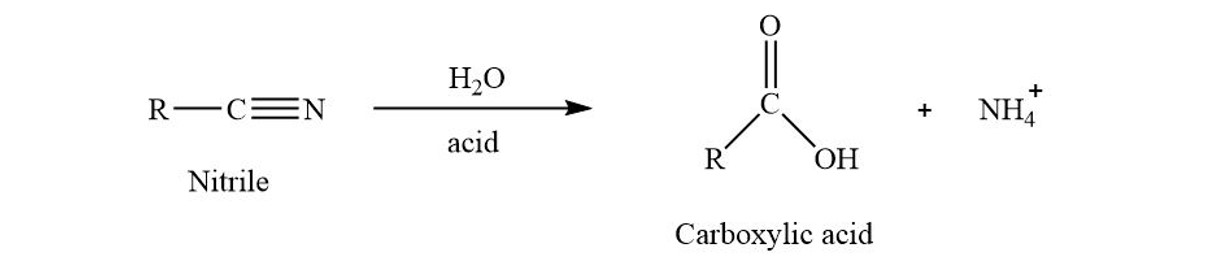
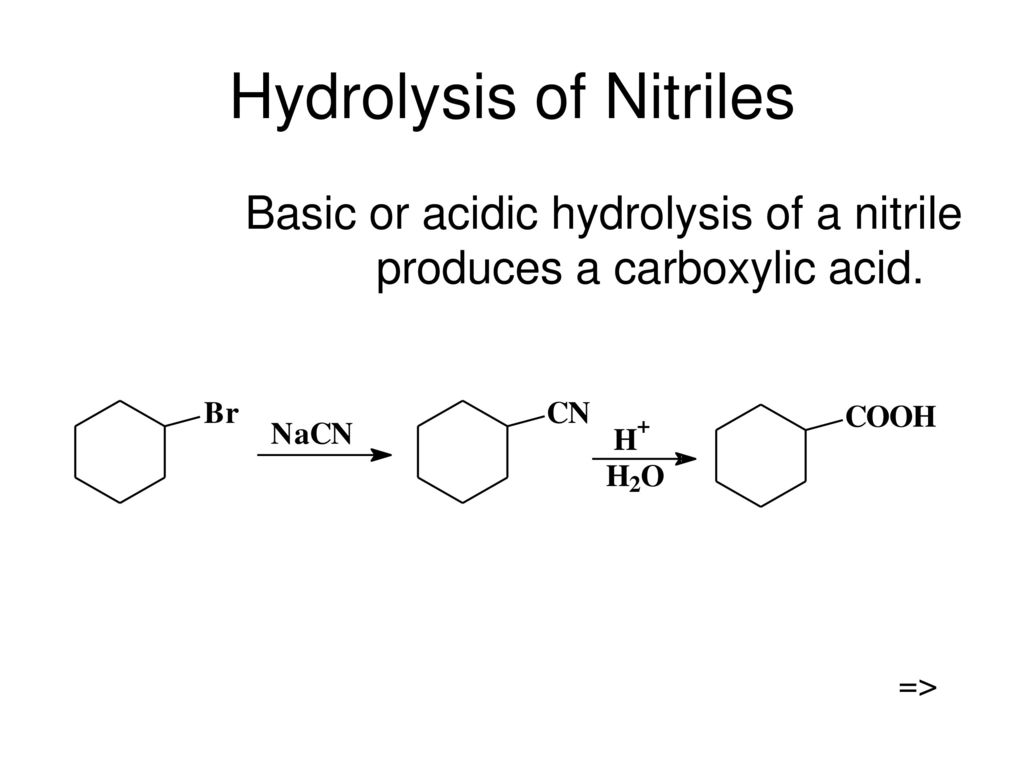
SOLVED: Cyanohydrins are nitrile intermediates in the synthesis of α-hydroxycarboxylic acids from ketones and aldehydes. The Schiobe Tydrokyile fiocica Group can be hydrolyzed by aqueous acid to yield carboxylic acid. Nitriles can