CoII immobilized on an aminated magnetic metal–organic framework catalyzed C–N and C–S bond for

Potassium Phosphate as a Solid Base Catalyst for the Catalytic Transfer Hydrogenation of Aldehydes and Ketones | ACS Catalysis

Synthesis of HTLcs modified by K3PO4 for side chain alkylation of toluene with methanol - ScienceDirect

Potassium Phosphate as a High-Performance Solid Base in Phase-Transfer-Catalyzed Alkylation Reactions | Industrial & Engineering Chemistry Research
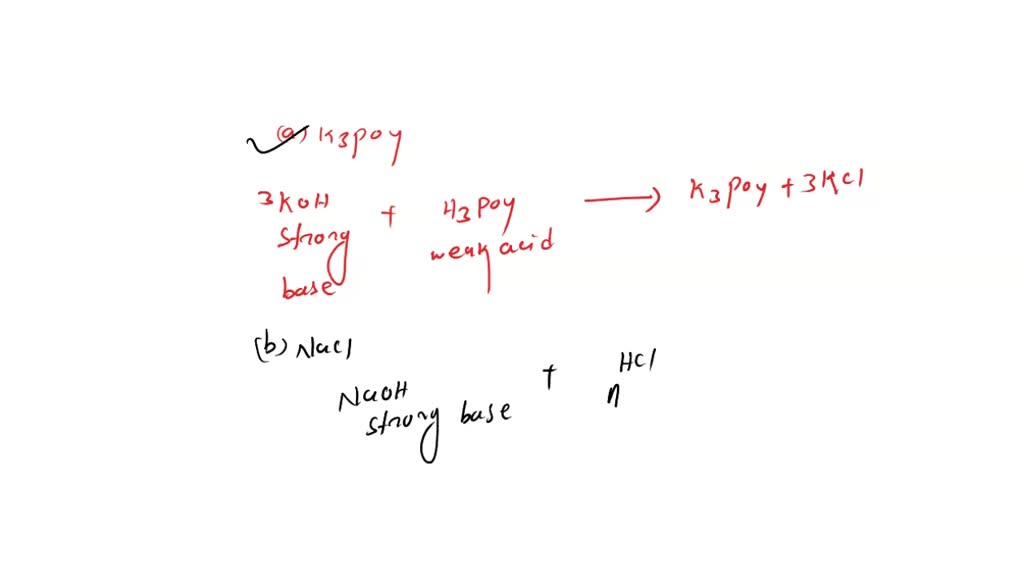
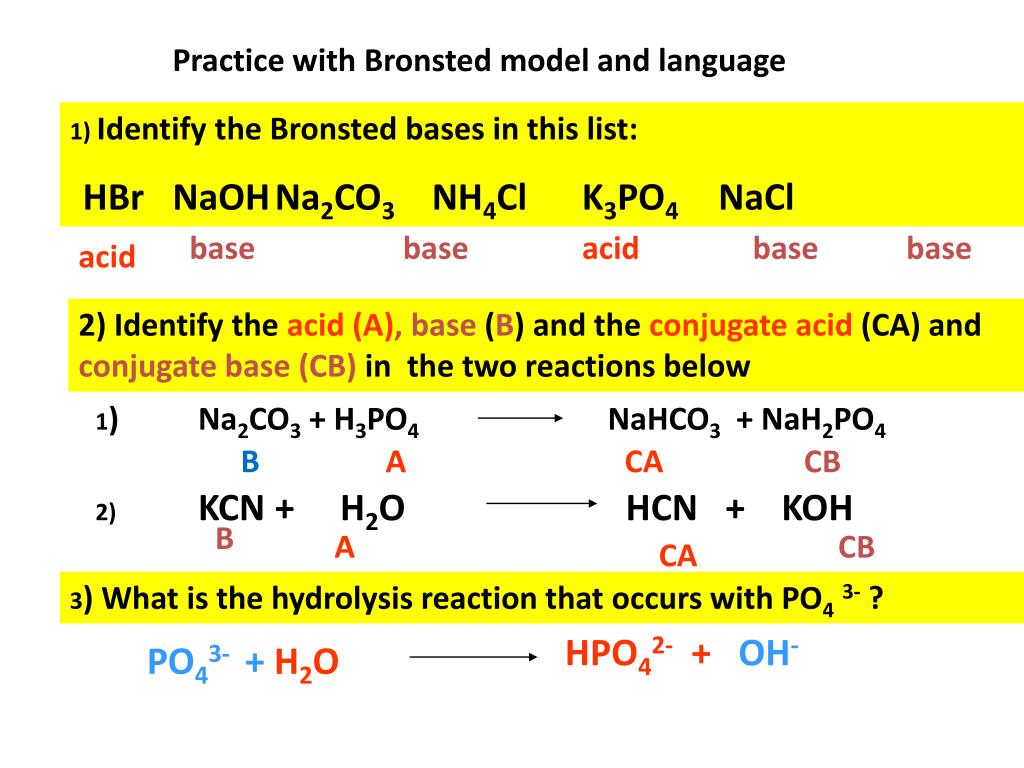
SOLVED: Which of the following salts may be obtained by the reaction of a weak acid with a strong base? a. K3PO4 b. NaCl c. FeCl3 d. LiClO4 e. NH4F

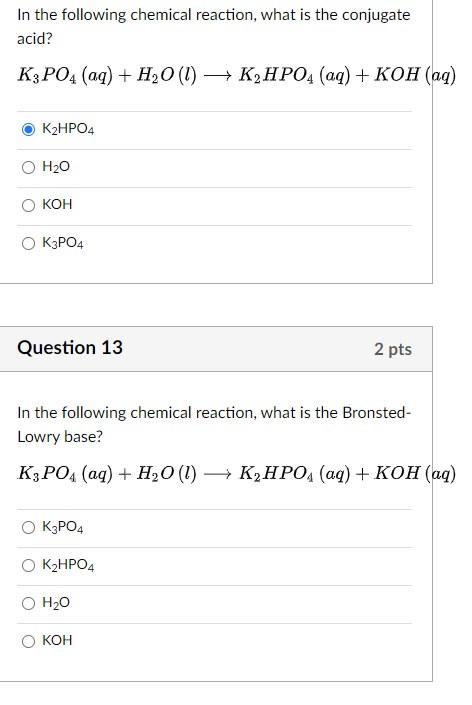
SOLVED: The balanced chemical equation for the reaction of the base potassium hydroxide with phosphoric acid to form potassium phosphate and water is: 3 KOH HPO4 KaPOs 3 HzO How many grams

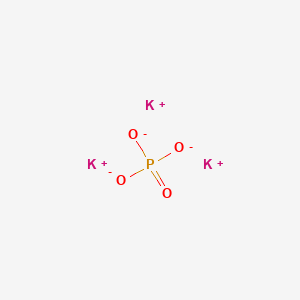
What is the formula of a compound that contains K^+ and PO_4^{3-} ions? (a) K_3P (b) K_2PO_3 (c) KPO_4 (d) K_3PO_3 (e) K_3PO_4 | Homework.Study.com

A Regioselective Synthesis of 6-Alkyl- and 6-Aryluracils by Cs2CO3- or K3PO4-Promoted Dimerization of 3-Alkyl- and 3-Aryl-2-Propynamides. | Semantic Scholar
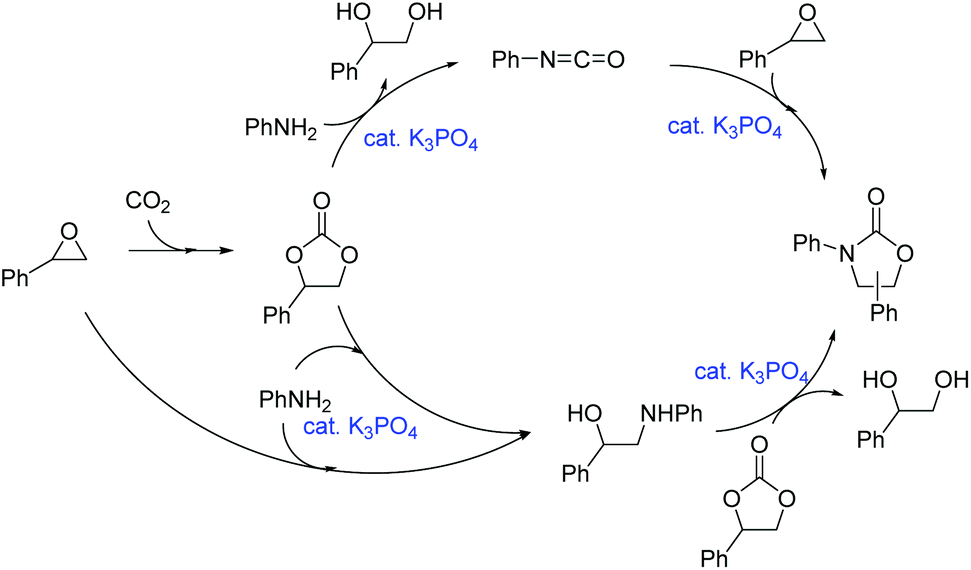
Potassium phosphate-catalyzed one-pot synthesis of 3-aryl-2-oxazolidinones from epoxides, amines, and atmospheric carbon dioxide - Green Chemistry (RSC Publishing) DOI:10.1039/C6GC02934E

Potassium Phosphate as a Solid Base Catalyst for the Catalytic Transfer Hydrogenation of Aldehydes and Ketones | ACS Catalysis