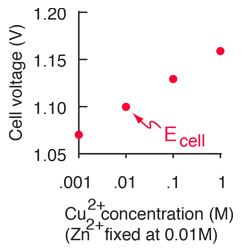
![Calculate the standard electrode potential of Cu^++ | Cu, if the electrode potential at 25^@C is 0.296V when the [Cu^++] = 0.015 M. Calculate the standard electrode potential of Cu^++ | Cu, if the electrode potential at 25^@C is 0.296V when the [Cu^++] = 0.015 M.](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/645222232_web.png)
Calculate the standard electrode potential of Cu^++ | Cu, if the electrode potential at 25^@C is 0.296V when the [Cu^++] = 0.015 M.

Question Video: Calculating a Cell Potential from Standard Electrode Potentials of Cadmium and Nickel | Nagwa

Why the standard hydrogen potential taken as reference for calculating standard electrode potential? - Quora

Question Video: Calculating the Standard Cell Potential for a Magnesium/Silver Galvanic Cell | Nagwa

Standard Cell Potential: Calculations, Electron Flow & Feasibility (5.4.2) | CIE A Level Chemistry Revision Notes 2022 | Save My Exams
Electrode configurations and reference electrodes Nernst equation: in principle possible to calculate and measure EMFs for half

Standard Reference Electrode Standard Hydrogen Electrode (SHE) SHE: Assigned V Can be anode or cathode Pt does not take part in reaction Difficult. - ppt download

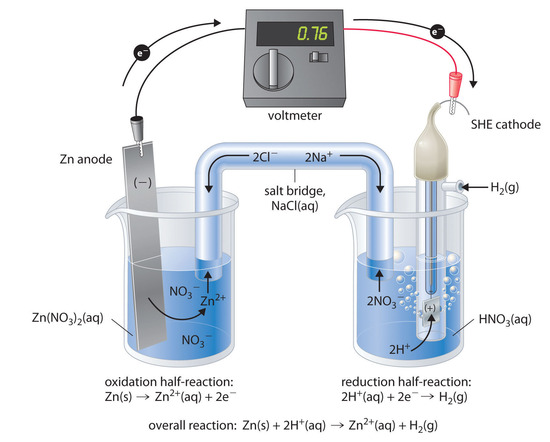
The standard electrode potential (E^∘) for Daniel cell is + 1.1 V . Calculate Δ G^∘ for the reaction. Zn(s) + Cu(aq)^2 + → Zn(aq)^2 + + Cu(s) (1 F = 96500 C/mol)

The standard electrode potential for Daniel cell is `1.1V`. Calculate the standard Gibbs energy - YouTube