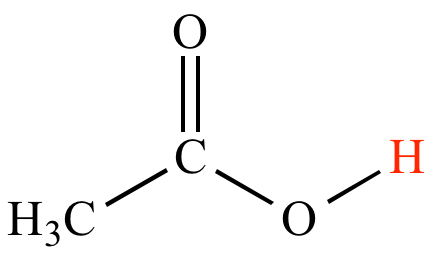
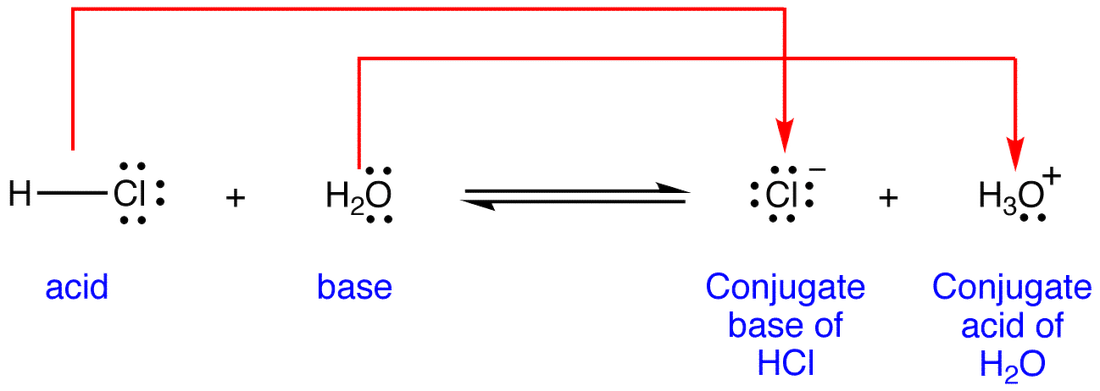
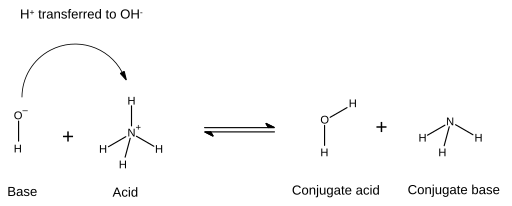
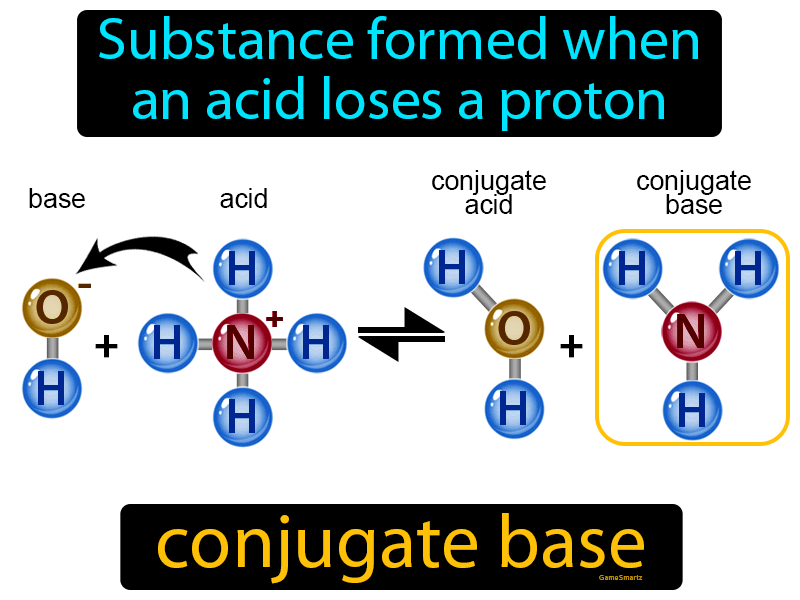
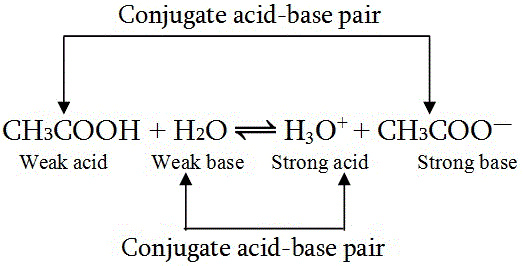
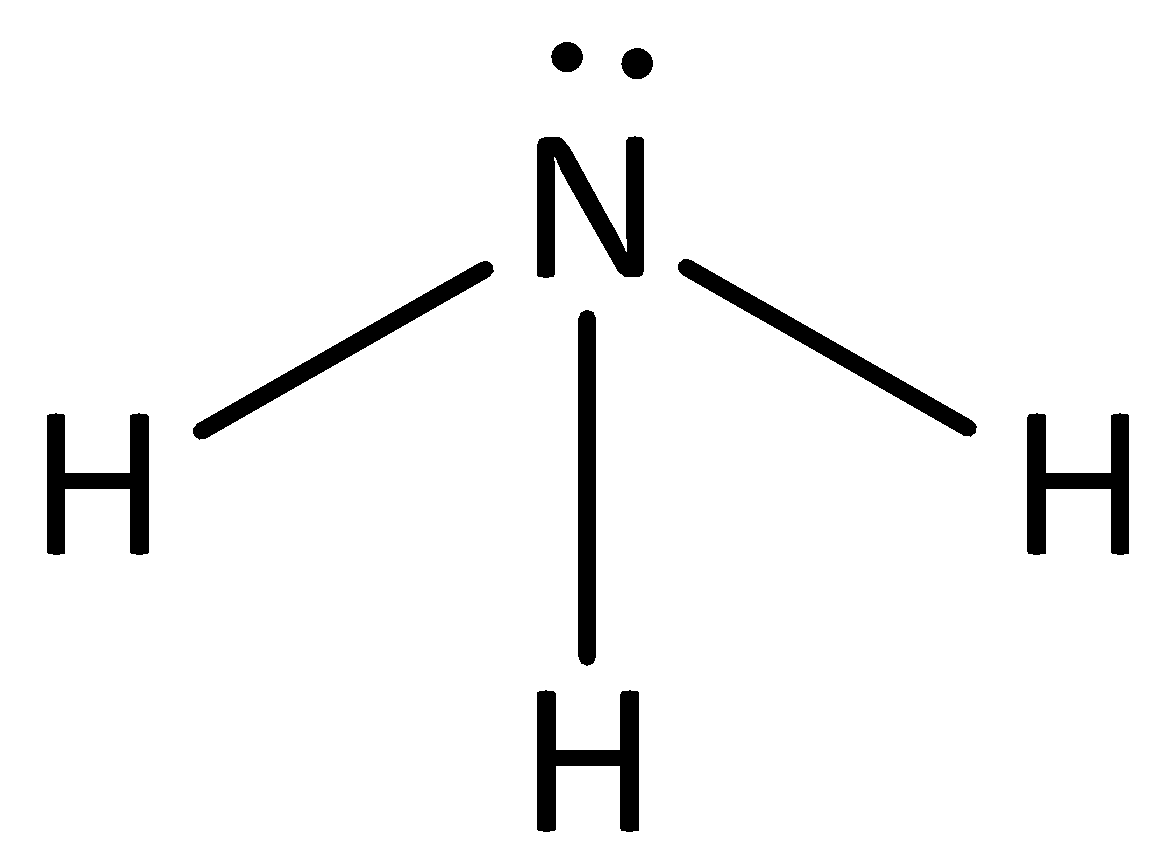
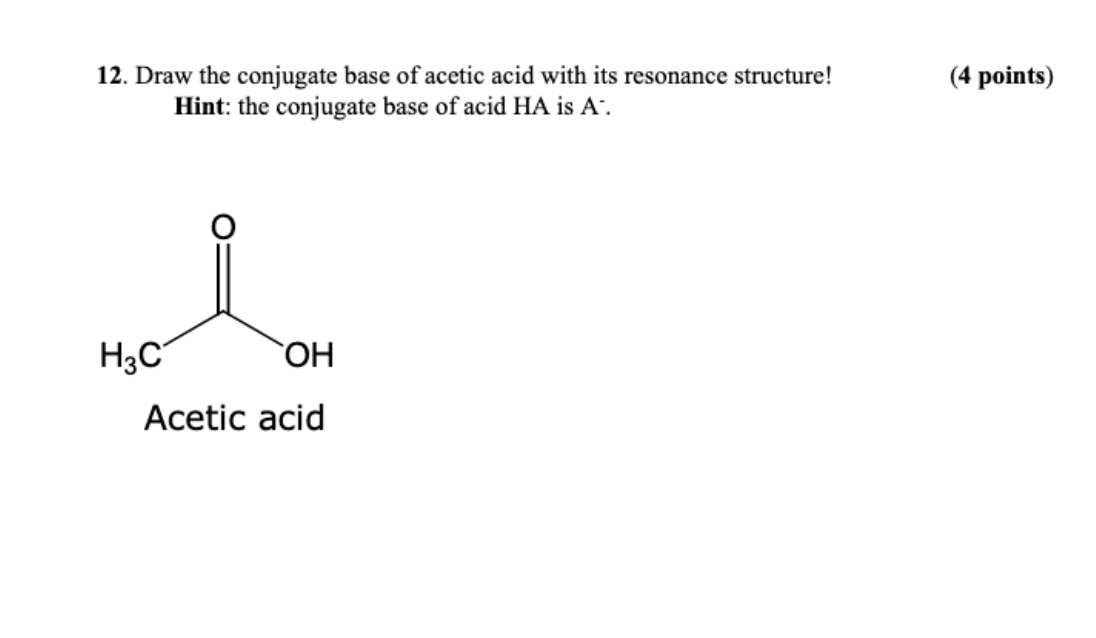
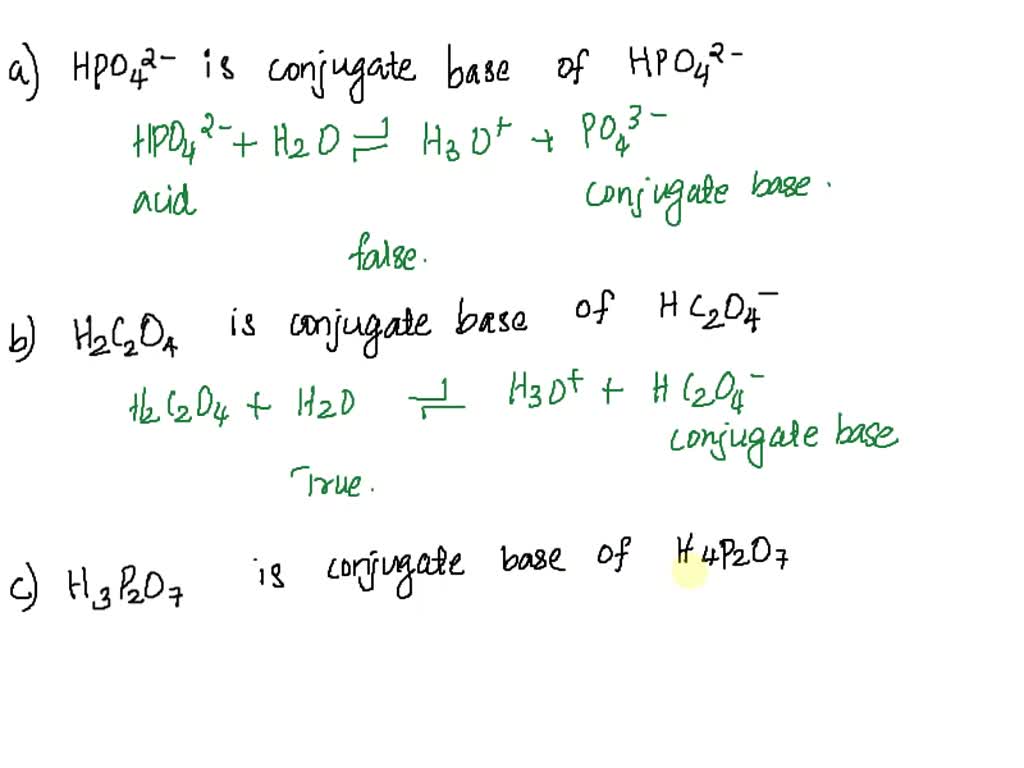Use your understanding of molecular structure to explain why the conjugate bases of acids like formic acid CHOOH, acetic acid CH3COOH, and phosphoric acid are only stable enough to be weak acids;

explanation 1) Look up the chemical name of aspirin and then its conjugate base (as the sodium salt) and provide those names here 2)The manufacturer's label for the "aspirin lists aspirin as

Use your understanding of molecular structure to explain why the conjugate bases of acids like formic acid CHOOH, acetic acid CH3COOH, and phosphoric acid are only stable enough to be weak acids;

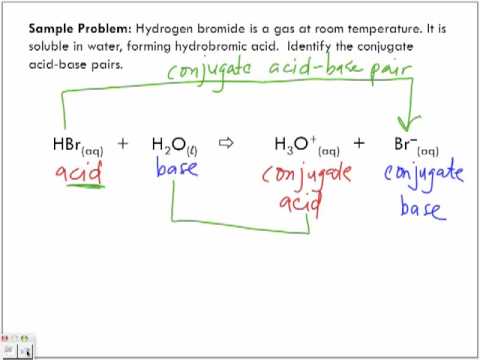
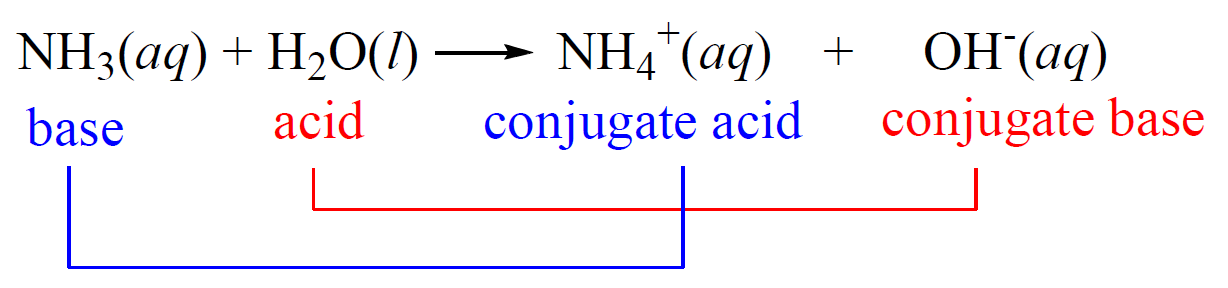
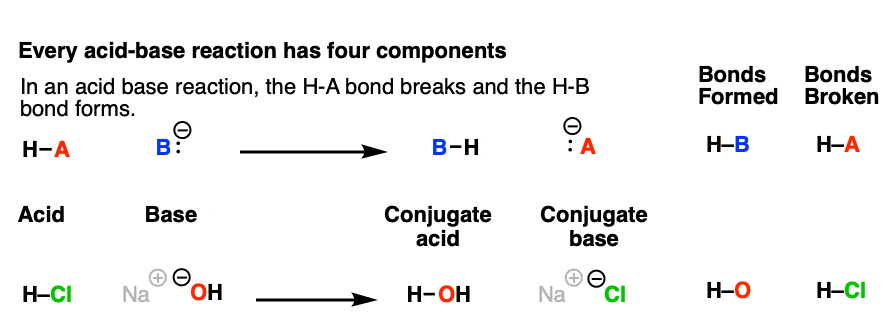
How to Identify Acid, Base, Conjugate Acid, and Conjugate Base Examples and Practice Problems - YouTube