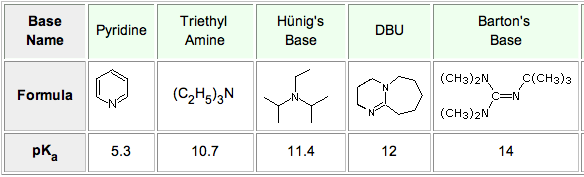
Synthesis of Highly Oxygenated Dinaphthyl Ethers via SNAr Reactions Promoted by Barton's Base | Organic Letters

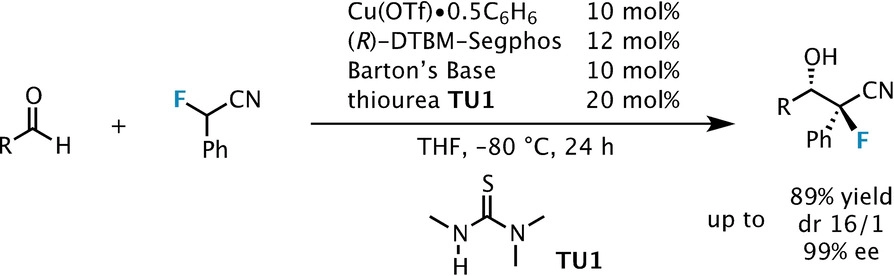
Catalytic asymmetric synthesis of CF 3 -substituted tertiary propargylic alcohols via direct aldol reaction of α-N 3 amide - Chemical Science (RSC Publishing) DOI:10.1039/C7SC00330G
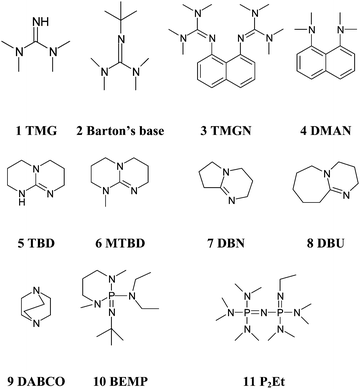
Kinetics screening of the N -alkylation of organic superbases using a continuous flow microfluidic device: basicity versus nucleophilicity - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB25215E

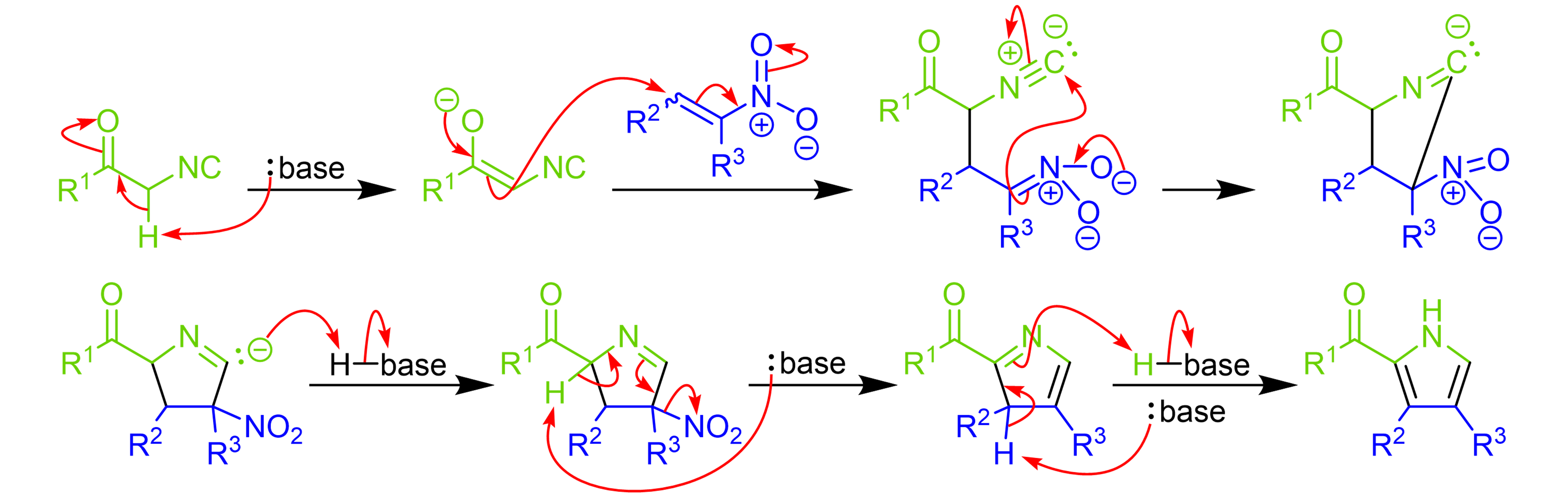
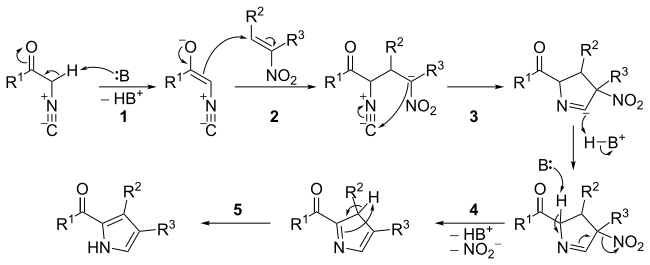
Direct Catalytic Asymmetric Mannich-Type Reaction en Route to α‑Hydroxy-β-amino Acid Derivatives - ScienceDirect

Buy Bartons Silver Plated Oval Base Decorative Basket (10 cm x 6.6 cm x 9 cm, Silver) Online at Low Prices in India - Amazon.in

Synthesis of Highly Oxygenated Dinaphthyl Ethers via SNAr Reactions Promoted by Barton's Base | Organic Letters
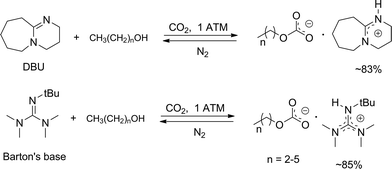
Anhydrous tertiary alkanolamines as hybrid chemical and physical CO 2 capture reagents with pressure-swing regeneration - Energy & Environmental Science (RSC Publishing) DOI:10.1039/C0EE00506A